Molecular Activity Measurement Applications
Measuring Molecular Activity with Optical Imaging
Optical imaging modalities are ideal for observing and characterizing biological and cellular processes in vivo with the right probe. These imaging modalities include detection using bioluminescence as well as fluorescence. Molecular activity can be measured by optical imaging either directly, using fluorescence optical probes or indirectly, using cells transfected with luciferase gene or iRFP.
NIR Optical Imaging Probes
Near-infrared (NIR) optical probes can be used to illuminate features within the animal (bone, vasculature, etc.). These probes may include specific targeting agents or non-specific contrast agents conjugated to near-infrared dyes.
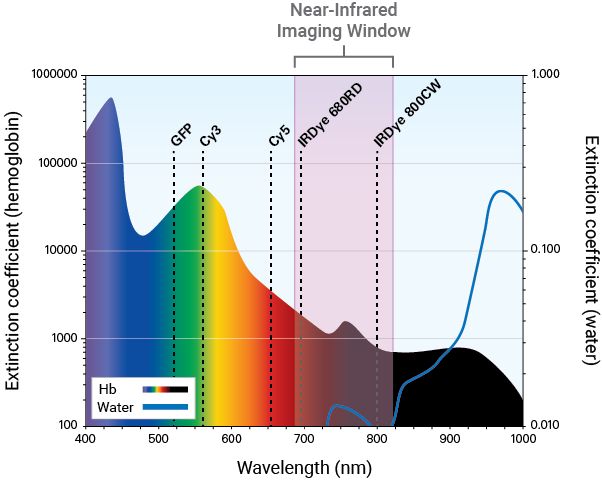
Detection in the NIR spectrum (680 nm to 820 nm) is ideal for measurement of in vivo molecular activity as it provides several advantages:
Sensitivity
Light absorbance by, and background autofluorescence from animal tissue is significantly lower in the NIR region. These properties allow better light penetration and higher signal-to-noise ratios for detecting deeper targets in vivo.
Throughput
Compared to other imaging modalities, images can be captured within seconds, allowing rapid screening of mice.
Cost
Compared to other modalities, optical imaging is more economical.
Bone Imaging
What is Bone Imaging?
Bone imaging is commonly used to provide anatomical landmarks to localize an area of tumor or disease. For instance, skeletal structures can be visualized using a dye-labeled bone targeting compound, while a second tumor-targeting agent labeled with a spectrally distinct dye, can help localize the tumor to bone anatomy. The two images can be overlaid to better define the location of the tumor or disease. Additionally, bone imaging in bone remodeling studies can detect bone growth or bone loss due to disease.
"…Investigators in the Vanderbilt Center for Bone Biology are using that probe [developed in Manning lab] to look at TGF‑beta in bone, and in and around tumors that may be in the bone, and that's been very fruitful with the Pearl."
Bone Imaging using NIR Fluorescence
BoneTag™ Optical Probes are calcium-chelating compounds conjugated to a near-infrared dye to enable detection of bone mineralization, growth, and morphological changes. BoneTag optical probes are available with the following near-infrared dyes: IRDye® 800CW (IRDye 800CW BoneTag) and IRDye 680RD (IRDye 680RD BoneTag). IRDye 680RD BoneTag probe can be imaged at the same time as another IRDye 800CW labeled targeting agent using the two-color imaging capabilities of the Pearl® Trilogy Imaging System.
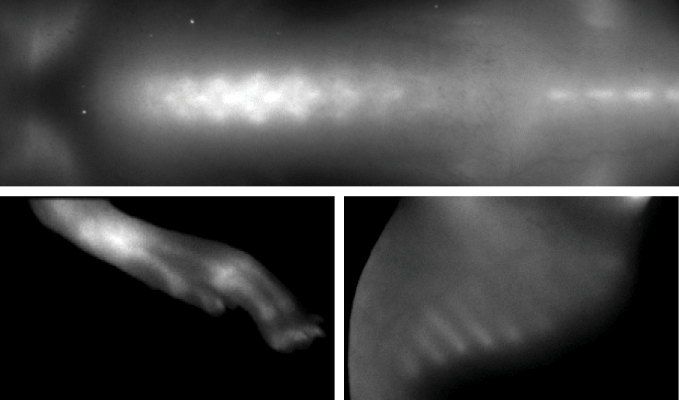
Figure 1. IRDye 800CW BoneTag was used to demonstrate resolution of structural imaging. Image captured with the Pearl Imaging System.
"…Investigators in the Vanderbilt Center for Bone Biology are using that probe [developed in Manning lab] to look at TGF‑beta in bone, and in and around tumors that may be in the bone, and that's been very fruitful with the Pearl."
Vascular and Lymphatic Imaging
What is Vascular and Lymphatic Imaging?
The vascular endothelium in the tumor microenvironment is often discontinuous, which allows molecules to diffuse into the surrounding tissue.1, 2 Lymphatic drainage in these regions is also poor.3 As a result, the tumor tissue is permeable to large molecules, and retains these molecules. Because of these properties, tumor vasculature and lymphatics can be studied using in vivo optical imaging probes.
Vascular and Lymphatic Imaging using NIR Fluorescence
IRDye 800CW dye is detected in the NIR spectrum where background autofluorescence from tissues is low. As a result, high signal-to-noise ratios are generated and provide images with excellent quality and resolution.
A near-infrared dye-conjugated contrast agent, such as IRDye 800CW PEG (polyethylene glycol), serves as a non-specific contrast agent for vascular imaging. The labeled agent is administered to mice intravenously (IV injection) and highlights surface vasculature for 30 minutes post-injection (Figure 2). The retention of the agent is visible in the tumor 4 hours post-injection (Figures 3A, B; requires appropriate mouse model*) and the tumor region is defined by 9 hours post-injection (Figure 4).
*Success of vascular imaging depends on the mouse model used. Vessels may be less visible in mice that are obese or have hair.
IRDye 800CW PEG also serves as an effective lymph tracking agent and can be administered by intradermal injection (Figure 5). Other contrast agents labeled with IRDye 800CW have been used in intraoperative identification of lymphatic branches and small sentinel lymph nodes, and have outperformed NIR quantum dots.4
The Pearl Trilogy Imaging System allows visualization of vasculature and lymphatics, without the need to change exposure settings between scans. Watch this two-minute video on rapid time-lapse lymphatic and lymph node imaging.
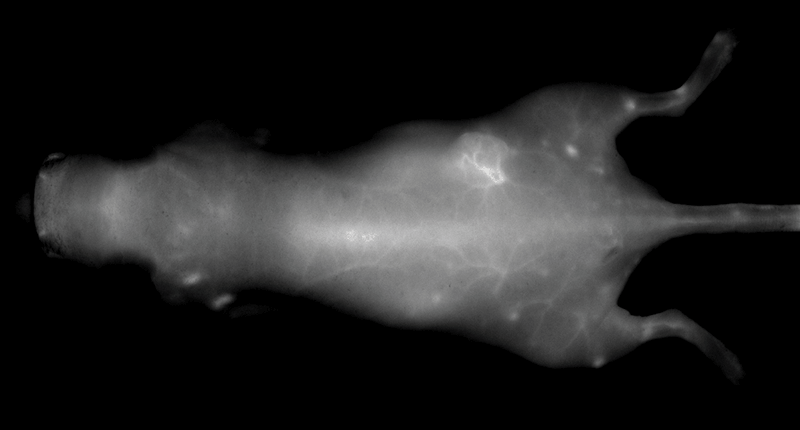
Figure 2. Athymic male nu/nu mouse (~5-6 wks old), 0.5 hr after injection of IRDye 800CW PEG (1 nmole). Surface blood vessels are visible. Note: ability to visualize vasculature is dependent on mouse model used. Image captured with the Pearl Imaging System.
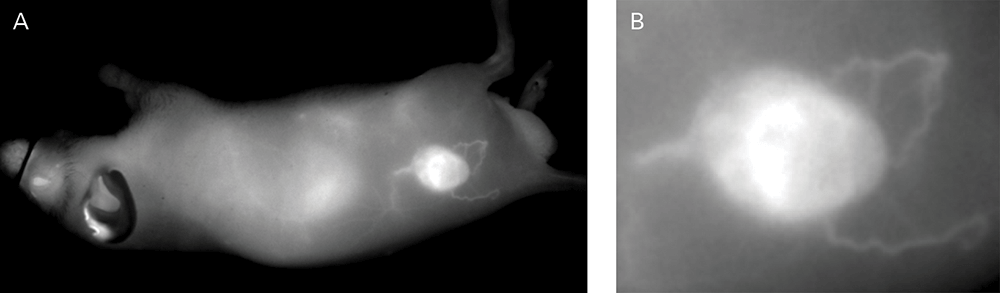
Figure 3. (A) Athymic male nu/nu mouse, ~4 hr after injection of IRDye 800CW PEG (1 nmole). Large blood vessels and tumor are visible. (B) High resolution (85 μm) image of the tumor region shows large blood vessels recruited to feed the tumor. Sequestration of contrast agent in tumor is likely due to enhanced permeability and retention. Image captured with the Pearl Imaging System.
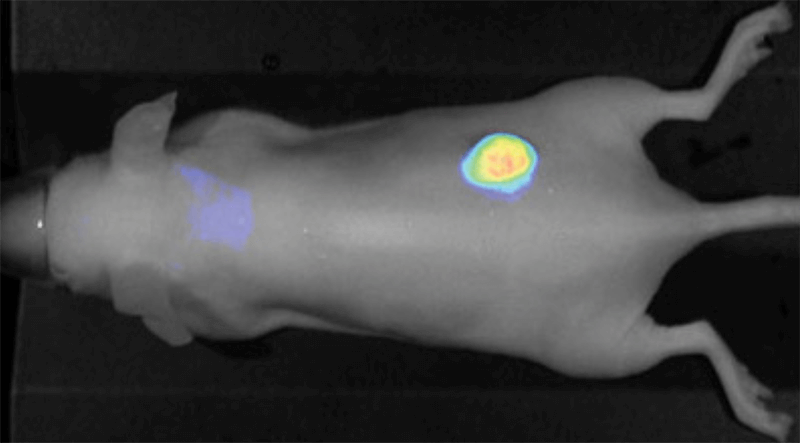
Figure 4. Athymic male nu/nu mouse, ~9 hr after injection of IRDye 800CW PEG (1 nmole). Tumor is clearly defined. Image captured with the Pearl Imaging System.
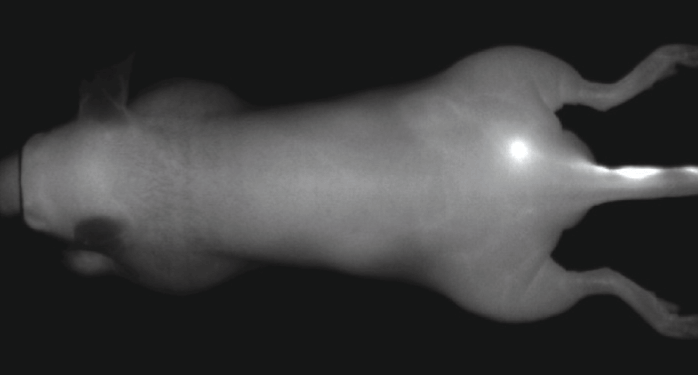
Figure 5. Athymic male nu/nu mouse, minutes after receiving IRDye 800CW PEG (~0.1 nmole) intradermally on the tail (right side). Image highlights use of IRDye PEG as a lymph imaging agent. Image captured with the Pearl Imaging System.
Measuring Molecular Activity with Transfected Cells
Molecular activity can be measured using transfected cells or transgenic animals expressing either the luciferase gene (i.e., bioluminescence) or fluorescent proteins like infrared Red Fluorescent Protein (iRFP) (i.e., fluorescence). With specific promotors, these genes can be used to monitor levels of expression—either constitutive or inducible—within an animal in context of the disease being studied.
Using Bioluminescent Proteins for Detection and Imaging
Bioluminescence is generally used for cell tracking and gene expression studies. Renilla luciferase and Firefly luciferase are the most commonly used enzymes in bioluminescence research. These enzymes oxidize the bioluminescent substrate luciferin, which results in an emission of light that can then be measured. Cells that constitutively express the luciferase gene can be injected into a mouse and followed over time for cellular distribution upon adding luciferin. Likewise, cells that express the luciferase gene under an inducible promotor can be monitored through various treatments to assess how the levels of expression change.
Bioluminescence can be detected using an imager like the Pearl® Trilogy Imaging System for in vivo molecular activity measurements. For example, a study compared D-luciferin and Cycluc-1 to determine which was a more robust luciferin substrate (Fig. 6). A cell-construct was implanted in mice, and xenografts were established. Upon the injection of substrate, the Pearl Trilogy Imager was then used to measure light production, and results suggested that Cycluc-1 was a more robust substrate than D-luciferin.
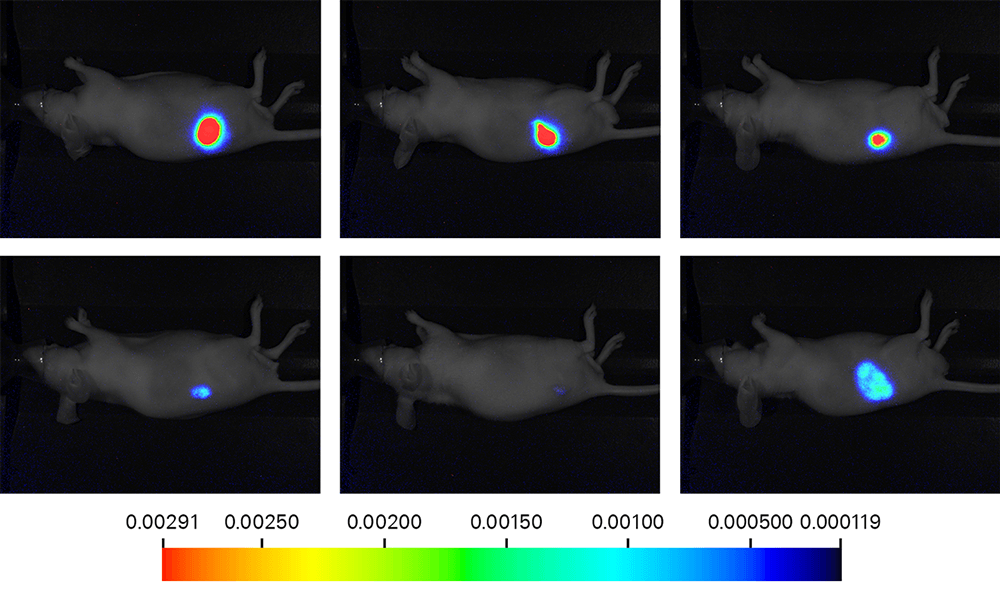
Figure 6. Mice were implanted subcutaneous in the right hind flank with cells that constitutively expresses luciferase. After 2 weeks, each mouse received 100 μL of 5mM D-luciferin or Cycluc-1 in PBS via intraperitoneal injection. The xenografts were measured by calipers to normalize signals to their volume. Volumes ranged from 62.6 – 691.2 mm3. Images were acquired on a Pearl Trilogy Imager. Research conducted at LI-COR Biosciences. (2016).
Fluorescent Protein Expression & Detection
Near infrared (NIR) fluorescent protein expression can be used to measure cell tracking and tumor progression. Unlike bioluminescence, iRFP does not require the addition of a substrate for visualization but simply needs to be excited using a light source. The emitted light is then detected by an imaging system such as the Odyssey® M, Odyssey DLx, or Pearl Trilogy.
Detecting Cancer Cell Lines & Monitoring Tumor Growth with iRFP
For in vivo molecular activity measurements, fluorescent protein expression can be detected using the Pearl Trilogy. For example, in a study by Henry et al., iRFP was used to detect the prostate cancer cell line 22Rv1 and monitor tumor growth in mice (Fig. 7). Images were then captured with NIR fluorescence and bioluminescence on the Pearl Trilogy imager.
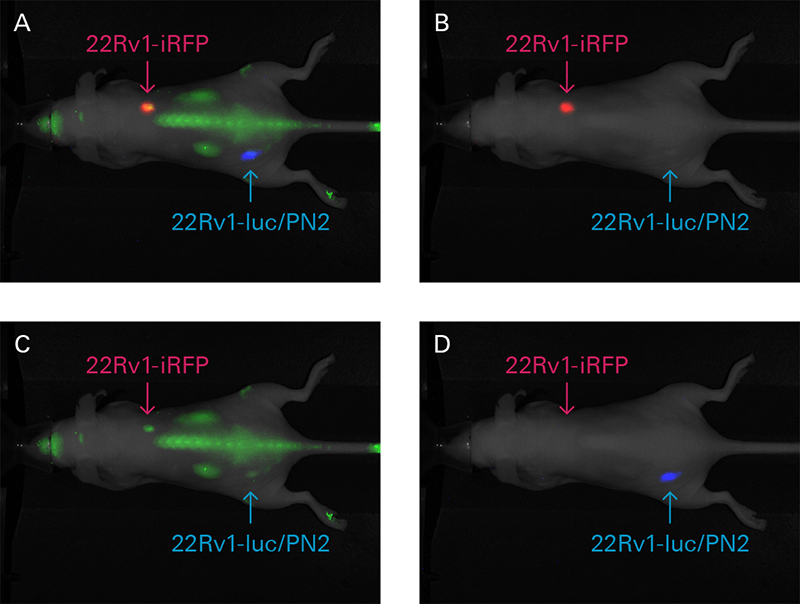
Figure 7. Mouse xenografts from iRFP-22Rv1 or 22Rv1-luc/PN2 constitutively expressing cells were prepared. Luciferase-expressing cells were implanted subcutaneous into the lower left hind flank, and iRFP-expressing cells were implanted subcutaneous into the right shoulder. IRDye 800CW EGF probe (green) was administered intravenously (1 nmole) to detect EGF expression in vivo. Images of iRFP (red) and IRDye 800CW EGF expression were captured 48 hours post injection, while bioluminescence (blue) was captured 10 minutes post D-luciferin injection. Individual images were captured in the B) 700 nm, C) 800 nm, and D) bioluminescence channels. A) All three channels are displayed in a composite image. Image adapted from Henry et al. (2005).5
Detecting iRFP in Small Cell Populations & Monitoring Tumor Growth
Reporter proteins are frequently used to study the regulation of gene expression in vitro and in vivo. A reporter gene is placed under regulatory control of a promoter of interest. Upon expression, it produces either a luminescent or fluorescent protein that can be used to detect protein localization or measure expression levels. There are several fluorescent reporter proteins that are commonly used; iRFP713, for instance, is a near-infrared (NIR) fluorescent protein with high intracellular stability and brightness compared to other phytochrome-derived fluorescent proteins. It is non-toxic in mammalian cells. Upon excitation of light at 690 nm, a signal is emitted at 713 nm that can be detected and measured using an imager.6
The expression of an infrared fluorescent protein such as iRFP713 alleviates some of the issues seen with visible reporter proteins like GFP.7 NIR wavelengths allow for deeper tissue penetration of excitation light as well as lower background autofluorescence of cells and tissues. iRFP713’s properties and the benefits of NIR wavelengths make iRFP713 a valuable tool for researchers wanting to study protein expression in both cells and live animals.
A study by Hock et al., detected in vivo iRFP713 expression and confirmed its sensitivity in a Drosophila model with NIR fluorescence on an Odyssey Imager (Fig. 8). iRFP713 was then used to monitor tumor progression in a genetically engineered mouse model (GEMM) in vivo with the Pearl Trilogy imager (Fig. 9).
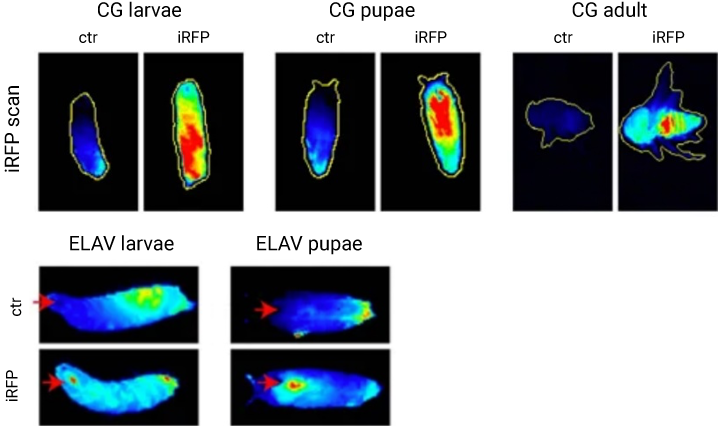
Figure 8. Detection of iRFP713 in vivo in a Drosophila model. iRFP713 was introduced to and detected in larvae, pupae, and adult CG driven Drosophila. iRFP713 expression in the central nervous systems of ELAV Drosophila larvae and pupae demonstrated that iRFP713 was detectable using drivers that were targeted to small cell populations. Images were acquired on an Odyssey imager, and data was quantified with Image Studio™ software. Image adapted from Hock et al. (2017).8
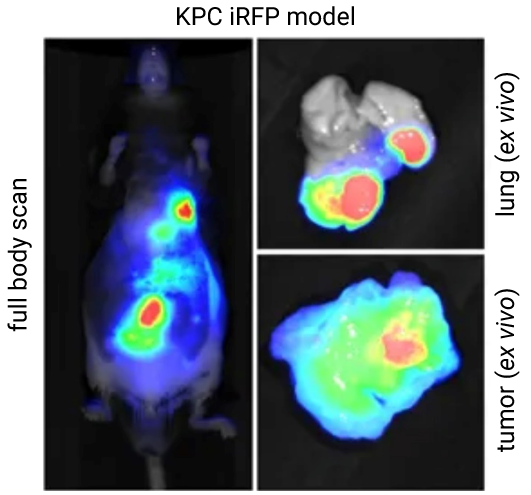
Figure 9. Using iRFP713 expression to track Pdx1-Cre activity and monitor tumor progression in vivo and ex vivo in a GEMM’s lung, gut, and pancreatic tissue. Metastatic tumors and metastases were resected and imaged ex vivo to confirm tumor presence. Images were acquired on a Pearl Trilogy imager, and data was analyzed using Image Studio software. Image adapted from Hock et al. (2017).8
References
- Vasey, PA et al. (1999). Phase I Clinical and Pharmacokinetic Study of PK1 [N-(2-Hydroxypropyl)methacrylamide Copolymer Doxorubicin]: First Member of a New Class of Chemotherapeutic Agents- Drug-Polymer Conjugates. Clinical Cancer Research, 5(1), pp 83-94.
- Matsumura, Y and H Maeda. (1986) A New Concept for Macromolecular Therapeutics in Cancer Chemotherapy: Mechanism of Tumoritropic Accumulation of Proteins and the Antitumor Agent Smancs. Cancer Res, 46(12), pp 6387-92.
- Seymour, LW. (1992). Passive Tumor Targeting of Soluble Macromolecules and Drug Conjugates. Critical Reviews in Therapeutic Drug Carrier Systems, 9(2), pp 135-87.
- Tanaka, E et al. (2006) Ann Surg Oncol, 13(12), pp 1671-81. DOI: 10.1245/s10434-006-9194-6.
- Drake, JM, Gabriel, CL & Henry, MD. (2005). Clin Exp Metastasis, 22(8), pp 674-84. DOI: 10.1007/s10585-006-9011-4.
- Filonov, G. S., Piatkevich, K. D., Ting, L.-M., Zhang, J., Kim, K., & Verkhusha, V. V. (2011). Bright and stable near-infrared fluorescent protein for in vivo imaging. Nature Biotechnology, 29(8), 757–761. https://doi.org/10.1038/nbt.1918
- Hock, A. K., Cheung, E. C., Humpton, T. J., Monteverde, T., Paulus-Hock, V., Lee, P., McGhee, E., Scopelliti, A., Murphy, D. J., Strathdee, D., Blyth, K., & Vousden, K. H. (2017). Development of an inducible mouse model of iRFP713 to track recombinase activity and tumour development in vivo. Scientific Reports, 7(1), 1837. https://doi.org/10.1038/s41598-017-01741-0
- Hock, A. K., et al. (2017). Development of an inducible mouse model of iRFP713 to track recombinase activity and tumour development in vivo. Scientific Reports, 7(1), 1837. https://doi.org/10.1038/s41598-017-01741-0
